|
Similar early clinical trials of CAR T cells in solid tumors in the 1990s using first generation CARs targeting a solid tumor antigens such as MUC1 did not show long-term persistence of the transferred T cells or result in significant remissions. Ī first generation CAR containing a CD4 extracellular domain and a CD3ζ intracellular domain was used in the first clinical trial of chimeric antigen receptor T cells by the biotechnology company Cell Genesys in the mid 1990s, allowing adoptively transferred T cells to target HIV infected cells, although it failed to show any clinical improvement. This work prompted CD3ζ intracellular domains to be added to chimeric receptors with antibody-like extracellular domains, commonly single-chain fraction variable (scFv) domains, as well as proteins such as CD4, subsequently termed first generation CARs. In 1991, chimeric receptors containing the intracellular signaling domain of CD3ζ were shown to activate T cell signaling by Arthur Weiss at the University of California, San Francisco. Originally termed "T-bodies", these early approaches combined an antibody's ability to specifically bind to diverse targets with the constant domains of the TCR-α or TCR-β proteins. at the Institute for Comprehensive Medical Science in Aichi, Japan, and independently in 1989 by Gideon Gross and Zelig Eshhar at the Weizmann Institute in Israel. The first chimeric receptors containing portions of an antibody and the T cell receptor was described in 1987 by Yoshihisa Kuwana et al. These two cell types, called CD4+ and CD8+, respectively, have different and interacting cytotoxic effects, and it appears that therapies employing a 1-to-1 ratio of the cell types provide synergistic antitumor effects. The surface of CAR T cells can bear either of two types of co-receptors, CD4 and CD8. CAR T cells destroy cells through several mechanisms, including extensive stimulated cell proliferation, increasing the degree to which they are toxic to other living cells (cytotoxicity), and by causing the increased secretion of factors that can affect other cells such as cytokines, interleukins and growth factors. 
When they come in contact with their targeted antigen on a cell's surface, CAR T cells bind to it and become activated, then proceed to proliferate and become cytotoxic. Īfter CAR T cells are infused into a patient, they act as a "living drug" against cancer cells. For safety, CAR T cells are engineered to be specific to an antigen that is expressed on a tumor but is not expressed on healthy cells. Once isolated from a person, these T cells are genetically engineered to express a specific CAR, which programs them to target an antigen that is present on the surface of tumors. ĬAR T cells can be derived either from T cells in a patient's own blood ( autologously) or from the T cells of another, healthy, donor ( allogeneically). Scientists harvest T cells from people, genetically alter them, then infuse the resulting CAR T cells into patients to attack their tumors. The premise of CAR T immunotherapy is to modify T cells to recognize cancer cells in order to more effectively target and destroy them. The receptors are chimeric in that they combine both antigen-binding and T cell activating functions into a single receptor.ĬAR T cell therapy uses T cells engineered with CARs to treat cancer. In biology, chimeric antigen receptors ( CARs)-also known as chimeric immunoreceptors, chimeric T cell receptors or artificial T cell receptors-are receptor proteins that have been engineered to give T cells the new ability to target a specific antigen. Engineered T cells are infused back into the patient Engineered T cells are expanded in tissue cultureĥ. 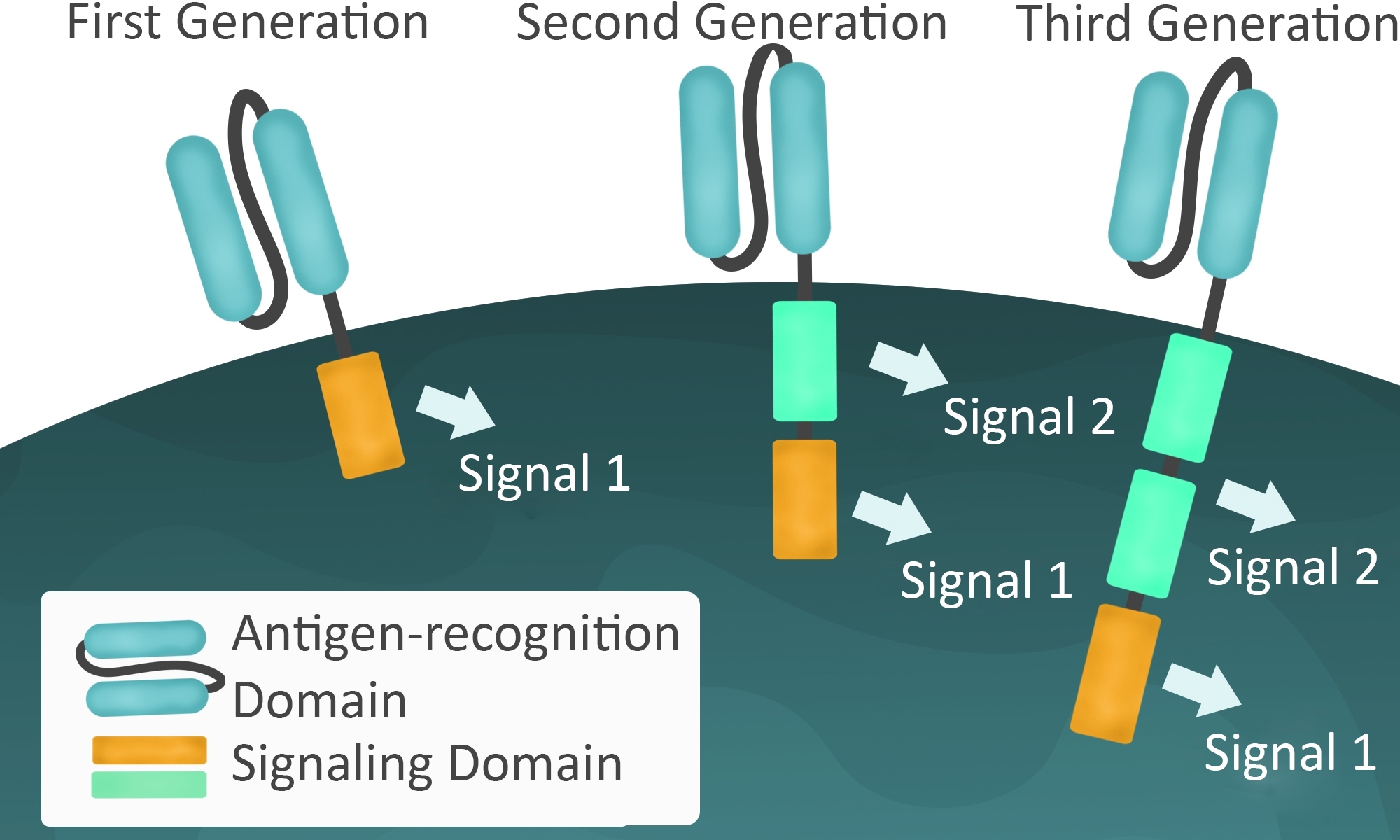 
Engineered T cells are now specific to a desired target antigenĤ. A new gene encoding a chimeric antigen receptor is incorporated into the T cellsģ. T cells are isolated from a patient's bloodĢ. Chimeric antigen receptor T cell production and infusion:ġ.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
